I’m taking the instrument check ride next month and I’m going to be blogging nightly on different topic in preparation for all of the stuff I think will come up on the oral part. I am preparing for lots of aviation weather talk, so at least the next week will be over those topics. I will have to explain things like the difference between a stable and an unstable atmosphere, what standard temperature and pressure values are, and the two basic ways that fog may form.
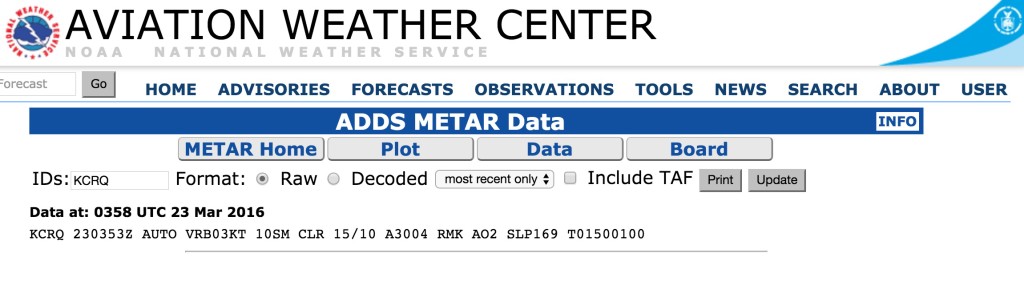
One very basic thing I will have to do is hop on aviationweather.gov and read and interpret a METAR. The first thing I will do is going to aviationweather.gov and click on METAR – it’s in the upper left hand corner of the website. Then I will scroll down to near the bottom of the page – on the right, where it says “Request Metar Data”, and I’ll type in the identifier of my airport – in this case KCRQ.
The METAR reads: Data at: 0358 UTC 23 Mar 2016 KCRQ 230353Z AUTO VRB03KT 10SM CLR 15/10 A3004 RMK AO2 SLP169 T01500100
Here’s how I’d read this: Airport is KCRQ, Carlsbad, CA Time – 23nd of March at 03:58 Zulu Time. I convert the time and it’s the 22nd of march at 20:58 Pacific Daylight time. The AUTO, when it appears just after the time group, means that the observation is from an automated station. Winds are light and variable at 3 knots, visibility is 10 or more statute miles, sky is clear, Temperature is 15 degrees, Dew Point is 10 degrees. Remarks: A02 means the station has a precipitation discriminator. SLP stands for Sea Level Pressure, which is another measure of atmospheric pressure. The Last digits that start with a “T” are the he hourly air and dewpoint temperatures to the nearest 1/10 C degree.
Tomorrow night I think I will review the Winds and Temperature aloft chart, and what valuable information can be determined on there, like the most favorable altitude, areas of possible icing, temperature inversions, and turbulence.









 The US Government Accountability Office
The US Government Accountability Office